Compliance Challenges in Pharma Patient Support


Key Takeaways
Compliance Challenges in Pharma Patient Support
Pharmaceutical companies face increasing challenges in managing Patient Support Programs (PSPs) due to stricter regulations. These programs, aimed at assisting patients with chronic conditions, must comply with federal laws like the Anti-Kickback Statute (AKS), False Claims Act (FCA), and HIPAA, as well as evolving state privacy laws such as California's CCPA. Non-compliance can lead to severe financial penalties, as seen in recent enforcement actions like Teva Pharmaceuticals' $450 million settlement in 2024 for FCA violations.
Key compliance issues include:
- Federal Regulations: AKS prohibits financial incentives influencing drug choices; FCA penalizes fraudulent claims tied to kickbacks; HIPAA mandates strict data privacy and security.
- State Privacy Laws: Broader definitions of personal information under laws like the CCPA and Washington's My Health My Data Act impose additional obligations.
- Data Security: Updated HIPAA rules now require mandatory measures like encryption, multi-factor authentication, and regular system testing.
To navigate this complex landscape, companies are leveraging technology with built-in compliance safeguards, such as encryption and audit trails, and adopting clear data governance frameworks. Properly designed PSPs not only align with regulations but also improve patient outcomes, increasing treatment adherence and confidence.
Keys to designing a compliant patient assistance program (PAP)
sbb-itb-8f61039
The Compliance Landscape for Patient Support Programs
Let’s dive into the intricate regulatory framework shaping patient support programs (PSPs), building on earlier discussions about their challenges.
Pharmaceutical companies face a maze of federal and state regulations governing everything from financial aid to data privacy. Non-compliance isn’t just a slap on the wrist - it can lead to massive penalties. For example, in October 2024, Teva Pharmaceuticals agreed to pay $450 million to settle allegations under the False Claims Act (FCA). The government claimed Teva funneled kickbacks through third-party foundations to cover Medicare copays for Copaxone between 2006 and 2017. This arrangement allegedly allowed Teva to hike prices while insulating patients from the financial burden.
State privacy laws add another layer of complexity, often imposing stricter requirements than federal regulations. Companies must adapt their PSPs to meet these heightened standards.
Federal Regulations: AKS, FCA, and HIPAA
Federal laws like the Anti-Kickback Statute (AKS), the False Claims Act (FCA), and HIPAA are central to PSP compliance.
The Anti-Kickback Statute (AKS) prohibits payments or incentives designed to influence the selection of specific drugs or services covered by federal healthcare programs. This has a direct impact on how financial assistance programs are structured. In January 2025, the U.S. Court of Appeals for the Fourth Circuit ruled in Pharmaceutical Coalition for Patient Access v. United States that a proposed setup - where oncology drug makers exclusively funded subsidies for their own drugs through a 501(c)(4) organization - could violate the AKS. The court emphasized that even narrowly defined subsidies risk steering patients and increasing Medicare costs.
The False Claims Act (FCA) holds companies accountable when kickbacks lead to fraudulent claims submitted to federal healthcare programs. Penalties under the FCA can climb into the millions, as recent cases have shown.
HIPAA governs how Protected Health Information (PHI) is handled in PSPs. To comply, modern systems often include encryption, role-based access, and audit trails. Beyond HIPAA, many programs follow additional security frameworks like SOC 2 and ISO 27001 to ensure thorough data protection. Programs that involve direct patient contact also trigger pharmacovigilance obligations, requiring systems to track and report adverse events discovered during patient interactions. Digital platforms like PatientPartner showcase how compliance can be built into program design.
These federal rules provide a foundation, but state-level privacy laws push the boundaries even further.
State Privacy Laws and Their Impact
State laws, such as the California Consumer Privacy Act (CCPA), create broader compliance demands compared to federal HIPAA regulations. The CCPA, for instance, governs "personal information" that doesn’t necessarily qualify as PHI. This means PSPs must meet additional obligations, like providing California residents the right to know how their data is shared for direct marketing purposes.
Recent updates to the CCPA have raised the bar even higher. Regulations now mandate cybersecurity audits, risk assessments, and oversight of automated decision-making tools for businesses processing consumer data. These 2026 updates also require companies to prepare for stricter audits of their data-handling practices. Additionally, digital platforms must incorporate state-specific consent mechanisms, such as the "usprivacy" string, to track whether consumers have exercised their rights under the law. For programs using AI-driven tools, compliance now includes transparent algorithms and risk-based approval processes to align with evolving regulatory standards.
Navigating this landscape requires constant vigilance and a proactive approach to compliance, especially as state and federal requirements continue to evolve.
Managing Patient Data and Privacy Compliance
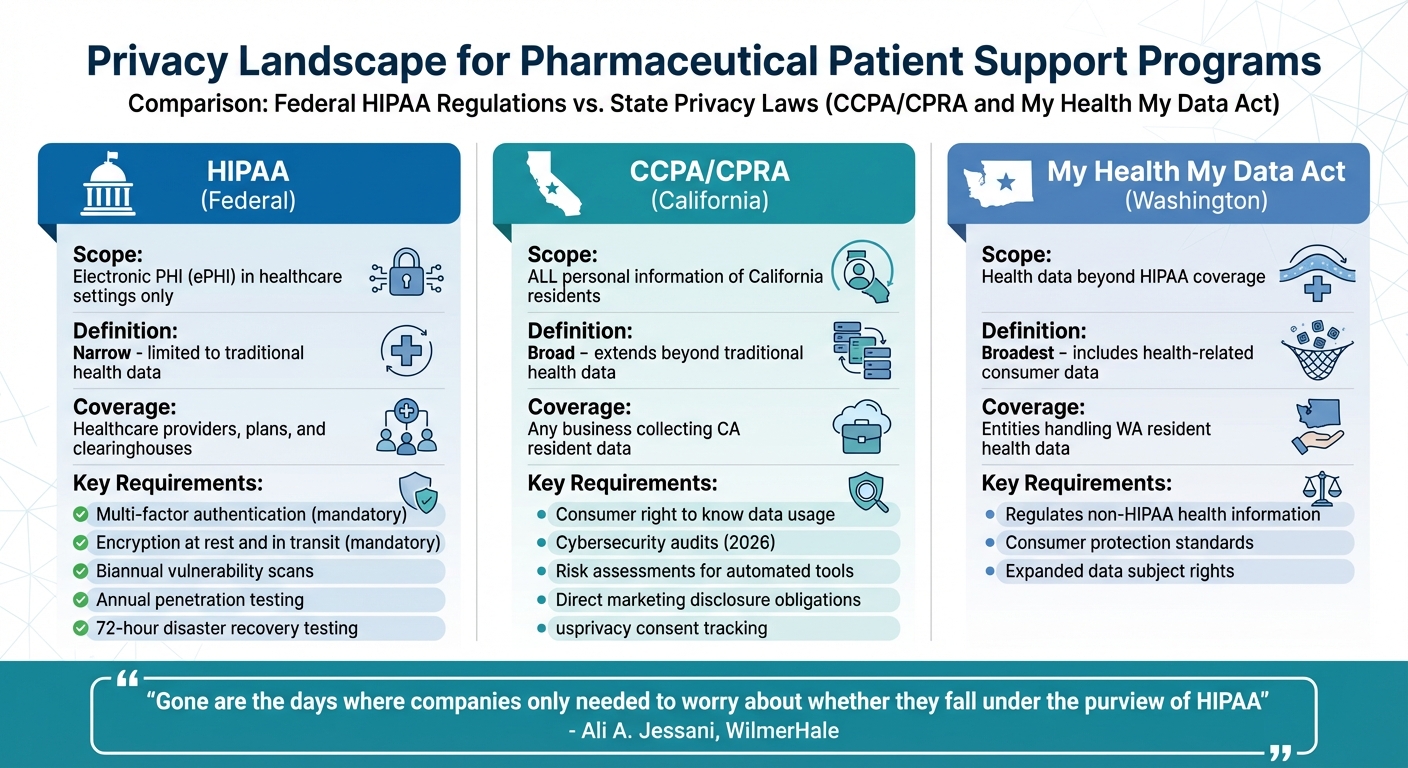
Federal vs State Healthcare Privacy Regulations Comparison Chart
Handling patient data today means navigating a maze of federal and evolving state regulations. The rules have become more complex, with overlapping and sometimes conflicting requirements. Let’s look at how differing definitions of protected health information (PHI) create hurdles for compliance across various jurisdictions.
Protected Health Information (PHI) Definition Challenges
HIPAA takes a fairly narrow approach to defining electronic PHI (ePHI), but state laws like California's CCPA/CPRA and Washington's My Health My Data Act cast a much wider net. This creates a compliance headache for organizations operating in multiple states.
Under HIPAA, ePHI is limited to electronic health data within healthcare settings. In contrast, California’s CCPA/CPRA governs all personal information of state residents, including data far beyond traditional health-related information. Washington's My Health My Data Act goes even further, regulating health data that HIPAA doesn’t even touch.
For pharmaceutical companies and others working across state lines, this patchwork of definitions is a major challenge. Ali A. Jessani, Counsel at WilmerHale, highlights this shift:
Gone are the days where companies only needed to worry about whether they fall under the purview of HIPAA.
Today’s regulations cover nearly any data that could be linked to health status, subjecting it to consumer protection laws even when it doesn’t meet HIPAA’s criteria for PHI. For example, companies serving California residents must juggle HIPAA's narrow scope with CCPA’s broader rules for personal information.
And while defining PHI is tricky, securing patient data comes with its own set of rigorous demands.
Data Security in Digital Patient Support Platforms
Recent updates to the HIPAA Security Rule have tightened the requirements for safeguarding ePHI. What were once "addressable" measures are now mandatory, leaving no room for leniency. As Johnson Lambert explains, regulators now expect companies to demonstrate strong security practices, rather than relying on good-faith efforts.
These mandatory measures include:
- Multi-Factor Authentication for all ePHI access points
- Encryption for data both at rest and in transit
- Biannual vulnerability scans and annual penetration testing
- Disaster recovery testing to ensure system restoration within 72 hours
For digital platforms facilitating peer-to-peer patient engagement, the stakes are even higher. These platforms must implement role-based access controls, maintain detailed audit logs of data interactions, and ensure secure, HIPAA-compliant communication tools to safeguard sensitive information during mentor-patient conversations. As PatientPartner emphasizes:
Compliance is at the core of everything we do. PatientPartner's platform is fully HIPAA and GDPR compliant, employing end-to-end encryption, role-based access controls, and audit trails to protect patient data.
Additionally, organizations must track their technology assets and maintain network maps that show exactly how ePHI flows through their systems.
The combination of evolving regulations and stricter security requirements makes managing patient data a complex but critical responsibility.
Recent Enforcement Actions and Settlements
Recent regulatory actions have highlighted critical vulnerabilities in patient support programs (PSPs). Even PSPs with the best intentions can face severe penalties if their programs are seen as influencing patient decision making. The Office of Inspector General (OIG) has taken a stricter stance on initiatives that could sway patients' drug selection, a position that federal courts have consistently upheld. Below are examples of how these regulatory positions have shaped enforcement actions.
Case Studies of Programs Falling Short on Compliance
In September 2022, the OIG issued Advisory Opinion 22-19, rejecting a program from the Pharmaceutical Coalition for Patient Access (PCPA). This program aimed to assist Medicare Part D cancer patients but was deemed to steer enrollees toward specific oncology drugs.
PCPA contested the OIG's decision in the U.S. District Court for the Eastern District of Virginia. However, in February 2024, the court ruled in favor of the OIG, reinforcing the agency's broad authority to interpret the Anti-Kickback Statute (AKS). As noted by Holland & Knight:
Healthcare compliance risks exist even when a company takes steps to structure its business activities to follow the government's own statements.
Another example comes from Advisory Opinion 20-05 (September 2020), where the OIG flagged direct cost-sharing assistance for a manufacturer's own drugs. The OIG warned that such programs could interfere with clinical decision-making and potentially increase healthcare spending by $32.3 billion annually.
In April 2019, two pharmaceutical companies faced hefty penalties, settling for nearly $125 million under the False Claims Act for similar violations.
Regulatory Guidance and Advisory Opinions
Not all OIG opinions have been unfavorable. For instance, Advisory Opinion 24-03 (June 2024) approved a nonprofit program managing 12 disease funds, each funded by a single manufacturer. These funds were structured around specific disease states, avoided promoting particular drugs, and supported non-drug costs like travel and lodging. However, the OIG limited the program's approval to December 31, 2026, citing concerns about evolving risks of fraud and abuse.
Other favorable opinions include Advisory Opinion 20-02 (modified June 2022), which permitted financial assistance for travel and lodging tied to cell-removal procedures for a specific drug. Similarly, Advisory Opinion 21-08 (July 2021) provided guidelines on when manufacturers can cover transportation, lodging, and meals for patients who might be eligible for their treatments.
Programs that focus on a wide range of drugs and prioritize non-drug-related costs tend to receive more favorable reviews. However, the PCPA case serves as a reminder that under the "one purpose test", a program can violate the AKS if even one of its purposes is to encourage drug purchases, regardless of whether the intent is corrupt - a principle upheld by the courts. These examples highlight the importance of PSPs building strong compliance frameworks to navigate these complex regulations.
Building a Compliance-Ready Patient Support Infrastructure
Pharmaceutical companies must weave compliance into the fabric of their patient support programs to avoid potential violations before they occur.
Using Technology for Compliance
Lessons from past enforcement actions have shown that advanced technology plays a key role in proactive compliance. Digital platforms now offer automated oversight and built-in safeguards. Take PatientPartner, for instance - it monitors patient-mentor interactions in real time to ensure compliance with HIPAA standards. George Kramb, CEO & Co-Founder of PatientPartner, emphasizes:
Compliance is at the core of everything we do.
PatientPartner's adherence to industry certifications such as HIPAA, SOC 2, and ISO 27001, along with its technical safeguards - like end-to-end encryption, role-based access, and audit trails - ensures compliance is embedded from the outset. These measures align with the 2025 updates to the HIPAA Security Rule, which have made safeguards like multi-factor authentication and encryption for data (both at rest and in transit) mandatory rather than optional.
The results speak for themselves: companies using PatientPartner have seen a 30% boost in treatment adoption. Patients stay on therapy an average of 133.5 days longer, are 68% more likely to start treatment, and report feeling 90% more confident in their decisions.
While technology provides a strong compliance foundation, maintaining adherence also requires ongoing policy measures.
Best Practices for Compliance Management
Technology alone isn't enough - clear policies and regular training are crucial for long-term compliance. Conducting a gap analysis is a good starting point, particularly to address the 2025 HIPAA updates that turn previously discretionary measures into mandatory requirements. It's also essential to verify that all partners meet annual certification standards.
Establishing a data governance framework is another important step. This includes creating a detailed data dictionary that outlines how data is collected, stored, and shared. Transparency in clinical decision support tools is equally important. Companies should allow independent reviews of recommendations and consult Institutional Review Boards (IRBs) to strengthen ethical oversight.
Conclusion
Pharmaceutical companies face a maze of federal and state compliance requirements when managing patient support programs. The stakes are enormous - medication non-adherence alone costs the U.S. an estimated $100 billion annually. On top of that, failing to meet privacy regulations can result in severe financial penalties and damage to a company's reputation.
To tackle these challenges, a well-rounded compliance strategy combines cutting-edge technology with strong policies, ongoing training, and clear oversight. Digital tools can automate safeguards to align with standards like HIPAA, SOC 2, and ISO 27001. When done right, compliant patient support programs yield tangible results: patients are 68% more likely to begin treatment, stay on therapy an average of 133.5 days longer, and feel more confident in their healthcare choices. These outcomes show that following regulations doesn't just reduce risks - it actively improves patient care.
By embedding compliance into their operations, pharmaceutical companies not only avoid pitfalls but also strengthen patient outcomes. As the industry shifts focus from "selling" to "serving", companies must position themselves as trusted partners, helping patients navigate their healthcare with privacy and security as top priorities.
At PatientPartner, compliance is the cornerstone of building trust and delivering patient-focused support.
FAQs
What compliance risks do pharmaceutical companies face when managing Patient Support Programs?
Pharmaceutical companies running Patient Support Programs (PSPs) face a range of compliance challenges due to strict regulatory standards from agencies like the FDA and the Office of Inspector General (OIG). Some of the biggest risks include improper communication with payers or healthcare providers, which could lead to accusations of misleading promotional practices or breaches of anti-kickback laws. PSPs also need to steer clear of any actions that might appear to influence prescribing decisions or offer financial incentives to patients inappropriately.
To navigate these challenges, companies must ensure that all patient engagement activities align with laws addressing fraud, abuse, and patient assistance programs. This means implementing strong compliance controls, providing clear and transparent disclosures, and following voluntary compliance guidelines. Taking these steps helps reduce the risk of penalties, reputational harm, and heightened regulatory scrutiny.
How do state privacy laws like the CCPA affect patient data management in support programs?
State privacy laws, like the California Consumer Privacy Act (CCPA), shape how patient data is managed in Patient Support Programs (PSPs). The CCPA mandates transparency, requiring organizations to disclose the types of data they collect, how they intend to use it, and with whom they share it. Patients are also granted specific rights, such as accessing their data, requesting its deletion, and opting out of data sharing or sales.
For PSPs, compliance with these laws means adopting robust privacy and security protocols. This involves creating clear, patient-focused data practices, protecting information from unauthorized access, and ensuring any data sharing strictly adheres to patient consent. Failure to meet these regulations not only risks legal penalties but can also damage an organization's reputation, making compliance a critical responsibility.
How can technology help ensure HIPAA compliance in patient support programs?
Technology plays a key role in keeping patient support programs in line with HIPAA regulations by safeguarding sensitive health information and ensuring privacy. Secure data management systems that are HIPAA-compliant, especially those using encryption both during storage and transmission, are critical for protecting patient data from unauthorized access.
Many software platforms now come with automated privacy policies and built-in safeguards, ensuring that every patient interaction adheres to regulatory requirements. Regular security risk assessments and maintaining detailed audit trails are equally important for tracking access to protected health information (PHI) and holding individuals accountable.
Another essential layer of compliance comes from educating staff. Dedicated training programs on HIPAA guidelines can help minimize human error and reinforce compliance efforts. On top of that, advanced patient engagement tools - like real-time mentorship platforms - can integrate privacy and security features right into their design, making it easier to meet HIPAA standards while improving patient care.
Author

Patrick Frank, Co-founder & COO of PatientPartner, leads healthcare patient engagement innovation through AI-powered patient support and retention solutions.


